UNDER THE MICROSCOPE
Bioengineering Assistant Professor George Alexandrakis is exploring how cancer cells damaged by radiation therapy work to repair themselves.

Cancer Crusaders
Breakthroughs in tissue engineering and optical imaging have brought UT Arlington bioengineers millions of dollars in funding to fight the disease that took more than 570,000 American lives last year.
· Spring 2012 · Comment ·
The numbers are alarming. The National Cancer Institute estimates that almost half the country’s male population will have some form of cancer, as will about one in three women. Though survival rates continue to improve, almost 35 percent of Americans diagnosed with cancer will die within five years.
Three UT Arlington bioengineering researchers aim to improve those statistics. Assistant Professors Baohong Yuan and George Alexandrakis and Professor Liping Tang have signed on as warriors against cancer, striving to develop more effective techniques to diagnose and combat the disease.
The Cancer Prevention and Research Institute of Texas (CPRIT) shares the trio’s enthusiasm for the potentially life-saving impact of their work, awarding them nearly $2 million in grants in 2011.
Dr. Yuan received $1.095 million to create a high-resolution imaging system that will detect small cancers in deep tissue. Dr. Alexandrakis, who secured a nearly $790,000 CPRIT award and a related $342,000 National Institutes of Health grant, focuses on how cancerous cells damaged by radiation therapy work to repair themselves. CPRIT awarded Dr. Tang, a tissue engineering expert, $200,000 to create a “chemical trap” that will mimic bone marrow and attract migrating cancer cells.
Texas voters approved CPRIT via constitutional amendment in 2007, authorizing $3 billion in bonds to fund cutting-edge cancer research and prevention programs and services. Fewer than 10 percent of the research applications typically are funded each year, with most of that going to medical schools.
“Receiving these CPRIT awards is a remarkable milestone for UT Arlington,” says Provost Ronald Elsenbaumer, who notes that bioengineering Assistant Professor Jian Yang earned a $200,000 CPRIT grant in 2010 for thyroid cancer research. “The funding puts our biomedical research on par with the best.”
SHARPENING THE FOCUS
Resolution is important to Yuan, particularly when it comes to one of his specialties: the ability to see small tumors deep within tissue.
ATTRACT AND CONQUER
Wayward cancer cells often cause tumors elsewhere in the body. Bioengineering Professor Liping Tang is creating a bone marrow-mimicking trap to attract these cells and confine them to a single location.

“The problem with current imaging of tumors is that what you get is not super clear but more like an out-of-focus photo,” he says. “If you think about regular micro-imaging methods, like ultrasound, there are fundamental limitations. For imaging of deeper tissues, you have to sacrifice resolution. This means if you want to see deep, deep tissue, you cannot see too small. There’s a trade-off between the imaging depth and the resolution.”
Perhaps not for long. Yuan’s research focuses on using ultrasound-mediated techniques, combined with microparticles or nanoparticles that tumors attract, to image small but deep tumors. Exposed to ultrasound waves, the particles become temporarily fluorescent and can be detected by a non-invasive probe system that he and other researchers are helping design.
The work is an application of biophotonics, an emerging area of scientific research that uses light and other forms of radiant energy to explore the inner workings of living organisms, enabling researchers to see, measure, analyze, and manipulate tissues in novel ways.
“You can see clearer, deeper, and smaller,” Yuan says. “It is extremely useful to spot small tumors before they grow. If a tumor grows beyond 1 or 2 millimeters, it has to generate new blood vessels to provide nutrients and oxygen. The imaging technique can tell you if that’s happening.”
And that information is useful in deciding treatment—surgery, radiation, even spot-chemotherapy.
“It’s going to be a big help in both diagnosis and developing therapy. The imaging is also precise enough to tell whether blood vessels supporting the tumor are developing or not after treatment, which tells the physician how effective the treatment is.”
Yuan’s research centers on designing and testing the imaging device, a process that should progress from examining artificial tissue in the laboratory to animal studies.
“The eventual goal, of course, is to end up with imaging success with people.”
A PROTEIN GLOW
Alexandrakis’ research world is both sub-cellular and fluorescent. He painstakingly observes protein exchange inside a highly magnified cancer cell, a slow process involving thousands of proteins. He tracks a single protein at a time with a unique perspective: The cells within which the protein exists have all been exposed to radiation cancer treatment.
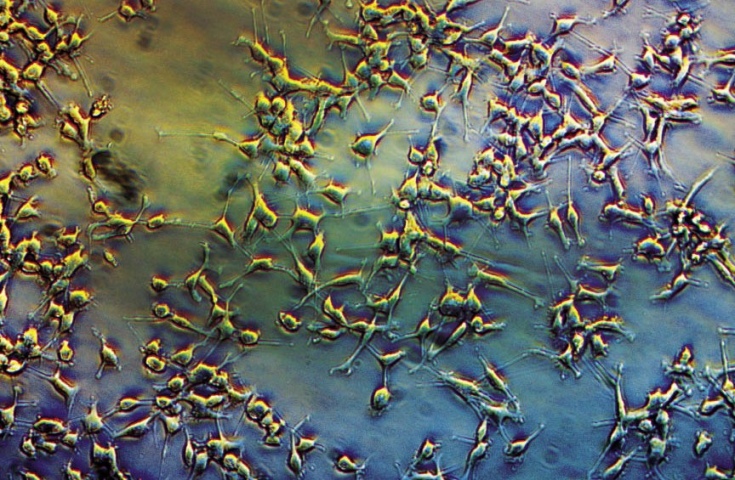
“We’re looking for ways to help cells repair themselves after radiation treatment,” says Alexandrakis, an imaging specialist with a doctorate in medical physics. He works with physicians and researchers at other institutions, including UT Southwestern Medical Center at Dallas and the UT MD Anderson Cancer Center.
“I’m the imaging guy who supports everybody else,” he says. “They ship me the cancer cells, I expose them to radiation treatment, and I tell them what’s actually happening in the cells, including how the healing process is going or not going.”
Evolving technology enables Alexandrakis to make a specific protein uniquely fluorescent within a cell.
“But if we can get those migrating cells to one spot, we will have a better chance to treat the cancer there and also to prevent it from spreading.”
“The cumulative interaction of multiple protein partners results in the DNA being repaired or in some cases mis-repaired. We try to break this down and understand all the steps in between as to how these proteins interact.”
In the long run, this should enable cancer treatment specialists to see how different variations of radiation and chemotherapy are working at a very early stage and to make adjustments.
Alexandrakis believes the research will progress from the cellular level to animal experimentation in about five years and from there to more refined and efficient treatment for human cancer patients. A prediction:
“With the knowledge of what’s happening within cells during radiation treatment protein interactions, all these different collaborations and these different research paths will eventually converge to more efficient and personalized treatment planning and to improved tumor destruction.”
IT’S A TRAP
From his state-of-the-art laboratory in the year-old Engineering Research Building, Tang examines a collection of special laboratory mice. They’re still alive months after being given a form of cancer that typically leads to death in about a month.
Each has been injected with a bioengineered, bone marrow-mimicking “trap” that attracts wayward cancer cells that would otherwise establish tumors elsewhere. Indeed, the mice without the traps have already died, most in fewer than five weeks.
“The test animals survive 25 percent longer with the implant trap alone,” Tang notes. “That’s without any other treatment whatsoever. Humans, of course, do receive treatment, which means that the trap has potential to both extend life or to help cure the disease entirely.”
IMAGE IS EVERYTHING
Bioengineering Assistant Professor Baohong Yuan and his team have received a grant for more than $1 million to create a high-resolution imaging system that will detect small cancers in deep tissue.

Cancer often kills by spreading, with cells migrating from an original tumor to other places in the body.
“Once that happens, the cancer can be hard to treat because it can be everywhere,” Tang says. “But some parts of the body have more attraction for migrating cells than others. Often we can’t stop the migration. But if we can get those migrating cells to one spot, we will have a better chance to treat the cancer there and also to prevent it from spreading.”
Bone marrow particularly attracts these potentially deadly agents. Tang’s research focuses on bioengineering a bone marrow replicant enhanced with one or more protein attractors that draw in cancer cells—for reasons not yet fully understood—like bees pulled to pollen.
It’s a two-step process. First the stem cell marrow mimicking is injected into the body. That’s the trap. Once researchers identify the factors that attract cancer cells to bone marrow (proteins that migrating cancer cells can sense), they can enhance the attraction.
Cancer that develops in the trap can be treated conventionally with radiation, chemotherapy, or surgery, and it’s far easier to treat a single site than multiple ones.
“So far this is looking like a pretty good strategy,” Tang says.
FOREFRONT OF DISCOVERY
That the research of Tang, Alexandrakis, and Yuan overlaps is no coincidence. Bioengineering Department Chair Khosrow Behbehani says that as early as 2002 the department decided to carve a niche in optical imaging and tissue engineering.
“Something that turned out beyond our expectation was that the medical optical imaging group has close collaboration with the tissue engineering group because they have complementary skills,” he says.
He’s thrilled at the University’s success in attracting bioengineering-related grants.
“We’re competing successfully with much larger institutions around the country. In particular, our decision to establish a UT Arlington imaging lab on the UT Southwestern campus has paid dividends. The idea was to make our researchers easily accessible to medical doctors and life science researchers. It is particularly valuable to invite somebody to our lab and show what we can do.”
Behbehani says the Holy Grail of cancer research is to understand what causes the disease at the cellular level.
“That’s going to be the direction, and understanding the essence is what is going to lead to more efficient ways to prevent or treat cancer. At UT Arlington we’re going to be on the front line of research leading to those accomplishments.”